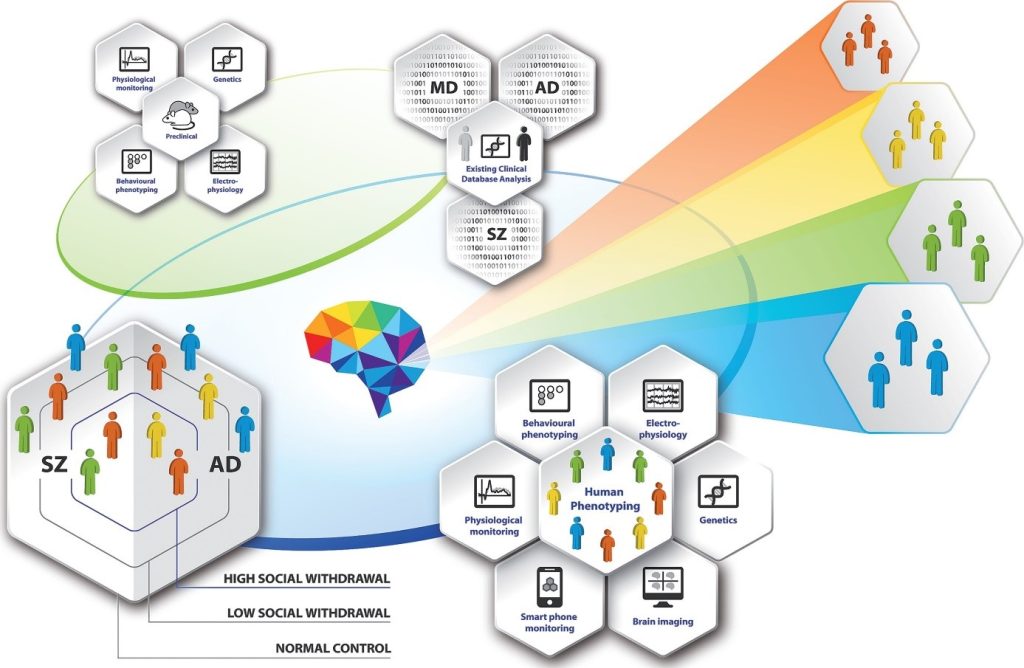
The current nosology of neuropsychiatric disorders provides a pragmatic approach to diagnosis and treatment choice, but lacks reference to quantitative biological underpinnings of disease. This weakness impedes innovative drug development. To test whether a quantitative biological approach to the understanding and classification of neuropsychiatric disorders is both feasible and useful the PRISM consortium was formed by academics, SMEs, patient organisations, regulators, ECNP, and EFPIA partners.
PRISM has successfully identified quantitative biological parameters related to diagnosis (Schizophrenia (SZ) and Alzheimer Disease (AD)) as well as to social functioning irrespective of diagnosis. From the relationships between social function, neuroimaging, and cognitive endpoints, a new neurobiological framework has emerged now, needing further validation. Genetic studies of social functioning outcomes revealed known and novel loci for this phenotype. In addition, a preclinical test battery was developed, based on homologs of the clinical paradigms, to allow effective back-translation and a deepening of our neurobiological knowledge. Finally, a novel digital tool for assessing social function provided a novel, objective characterisation that transcended the initial diagnostic classification and the digital readouts were associated with other study parameters.
To build on outcomes of PRISM, PRISM 2 has three objectives. First, to determine the reproducibility of the transdiagnostic and pathophysiological relationship between default mode network (DMN) integrity and social dysfunction in SZ and AD that emerged from PRISM and determine its potential to generalise to Major Depressive Disorders (MDD). Second, to test the causality between the quantitative variation in DMN integrity and social dysfunction. Third, to translate and communicate project results to the benefit of stakeholders, such as regulators, patients and their families, and health care providers.